Magnets steer medical microbots through blood vessels
Late one crisp October night in 2006, a hospital technician in Montreal slid the limp body of an anesthetized pig into the tube of a magnetic resonance imaging machine, or MRI. A catheter extended from a large blood vessel below its neck—a carotid artery. Into the catheter, a surgeon injected a steel bead slightly larger than the ball of a ballpoint pen.
In a room next door, my engineering graduate students and I held our breath. We were testing a program designed to manipulate the machine’s magnetic forces, which would guide the bead like a remote-controlled submarine. Or so we hoped.
On a computer screen, the bead appeared as a square white tracking icon perched on the gray, wormlike image of the scanned artery. We stared at the square and waited. Nothing. Seconds ticked by, and still the bead refused to budge. Then suddenly the room erupted in cheers as we clapped our hands to our mouths and pointed at the screen. There, the bead was hopping up and down the artery, tagging every waypoint we had plotted.
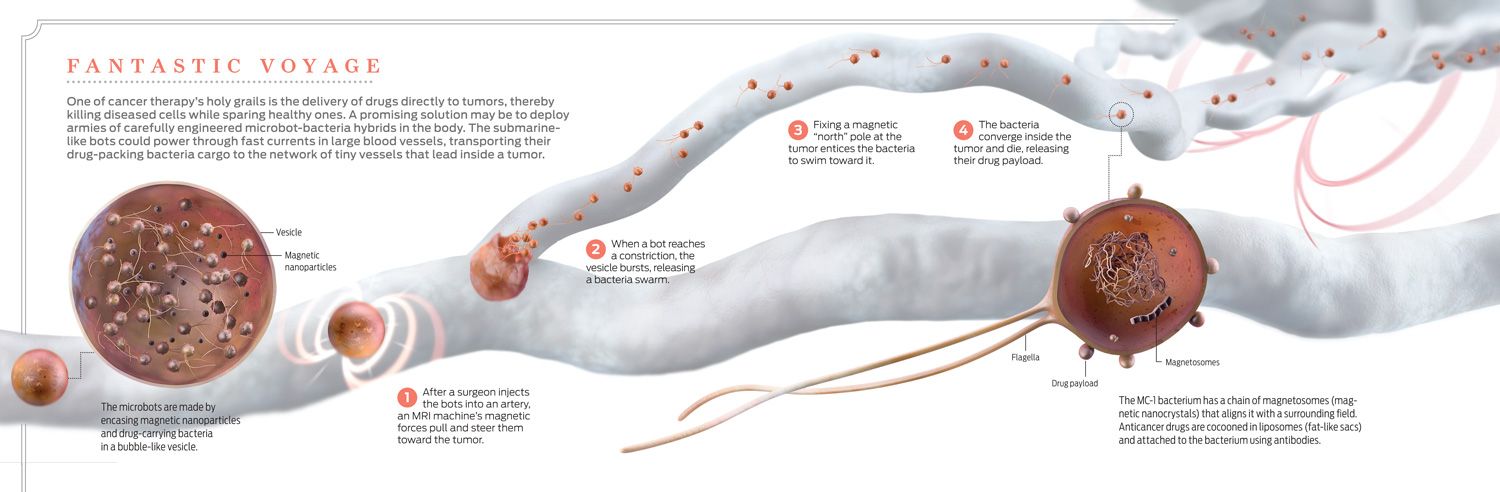
Illustration: anatomyblue
That was the first time anyone had steered an object wirelessly through the blood vessel of a living creature. The experiment convinced us that we could engineer miniature machines to navigate the vast circulatory system of the human body. The microrobots would be able to travel deep inside the body, cruising our tiniest blood vessels to places that catheters can’t go and performing tasks that would be impossible without invasive procedures.
It’s easy to imagine many such tasks—delicate surgeries, diagnostic tests, and the installation of stents and other artificial structures. But the first real application will be treating cancers. Today’s cancer drugs work by circulating throughout the body, killing healthy cells along with cancerous ones. Even antibody-equipped drugs designed to target cancer cells don’t always hit their marks. Injecting drugs into a tumor is out of the question because the pressure would cause cells to spew from it like a volcano, spreading the disease elsewhere. So why not deploy robots to deliver the medicine?
It’s a huge engineering challenge—one that will require input from many disciplines. The bots will have to contend with a complex maze of vessels churning with viscous blood. To make matters worse, today’s manufacturing techniques can’t build a motor and power source small enough to squeeze inside the capillaries—delicate vessels just a few micrometers thick—that feed a tumor. These vessels are too small for medical imaging tools to discern. Maneuvering through them would be like driving a car with your eyes closed.
Images: NanoRobotics Laboratory, École Polytechnique de Montréal MAKING A MEDICAL MICROBOT: Engineers encase medicine in liposomes and attach them to the bodies of bacteria, as shown in this scanning electron microscope image (top). They then load the bacteria into a vesicle or micelle sac (middle). To guide the bacteria-packed bot through an artery, they plot waypoints on a computerized image of the vessel (bottom).
Yet despite these challenges, my team at École Polytechnique de Montréal has made enormous progress on the problem. We have designed magnetic drug carriers as small as 50 micrometers that we can steer, like the metal bead inside the pig, through large arteries and arterioles using an MRI machine. To reach tumors deeper in the body, we need robots too small to be powered by technology. And so we have turned to nature, harnessing swarms of swimming bacteria to serve as drug mules.
There are many problems to solve and medical trials to do. But I believe the world will start to see cancer-fighting robots on the market within the next decade. One day, microbots may even be able to hunt and kill cancer cells before they grow into tumors.
Storytellers and scientists alike have long dreamed of miniature robots that can roam the human body, sniffing out disease and repairing organs. You may recall the old science fiction flick Fantastic Voyage (1966), in which a submarine and its crew are shrunk to the size of a microbe and sent into the bloodstream of an eminent scientist to destroy a blood clot in his brain. Physics Nobel laureate Richard Feynman shared a similar fantasy. In a classic 1959 speech, he described a friend’s “wild idea” that “it would be very interesting in surgery if you could swallow the surgeon.” As Feynman’s friend imagined it, a little mechanical surgeon marches through the bloodstream to the heart and surveys its surroundings. Upon spotting a problematic obstruction or a faulty valve, it simply “takes a little knife and slices it out.”
The problem, of course, is how to make the surgeon. Feynman envisioned an iterative manufacturing process in which ever-tinier hands make ever-tinier machines. He also considered the far-fetched dream of building things atom by atom, the way kids build toys out of Lego bricks. But he failed to offer any practical approaches. “I leave that to you,” he told his audience.
Four decades later, the closest engineers had come to realizing Feynman’s challenge was a diagnostic device called the PillCam. Introduced by the Israeli company Given Imaging in 2001, the disposable plastic capsule is about the size of a single piece of Good & Plenty candy and can be swallowed with water. It contains a light, a camera, batteries, an antenna, and a radio transmitter. As it passes through the gastrointestinal tract, it beams pictures wirelessly to a recording belt worn around a patient’s waist. The images are then analyzed for signs of cancers, Crohn’s disease, and other disorders. The procedure is less invasive than a traditional endoscopy and more sensitive than X-ray imaging.
But while the PillCam is great at inspecting the large, easily accessible cavities of the digestive system, it’s much too big to travel elsewhere in the body. The device is also passive. A true medical microbot must propel and steer itself through an intricate network of fluid-filled tubes to tissues deep inside the body. And because it is so small, it must do so without the convenience of a battery-powered motor.
You might think that an obvious way to accomplish this task is to equip the robot with metallic particles and guide it with a magnet from outside the body. You wouldn’t be entirely wrong. Indeed, some magnet-tipped surgical catheters work in this way. But a permanent magnet poses several problems for a smaller, tetherless robot. For instance, the magnet must be positioned quite close to the tiny particles to exert enough force to attract them. The robot could operate only near the skin and in one direction—toward the magnet. It would also be difficult to control. As it moved toward the magnet, the pull would increase, accelerating the robot and making it impossible to redirect if it went astray. And how would you know where it was going, anyway?
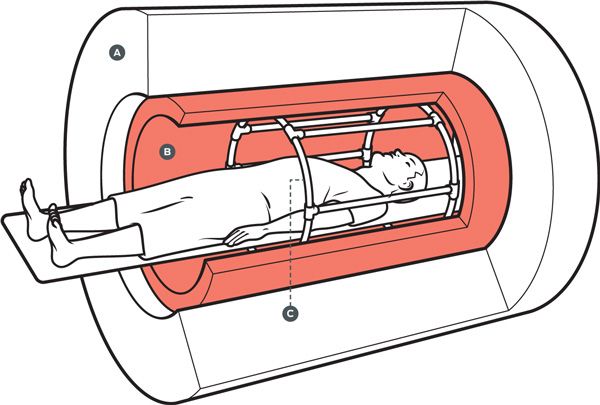
Illustration: James Provost INSIDE an MRI: To capture the body’s interior, a scanner uses three sets of electromagnetic coils: a multiton superconducting magnet [a], magnetic gradient coils [b], and a radio-frequency transceiver [c]. Click image to enlarge.
The answer is the MRI machine. To understand how an MRI machine can be used for robotic navigation, you must know something about how it works. The bulk of the machine is a powerful, doughnut-shaped superconducting magnet that generates a magnetic field up to about 60 000 times as strong as Earth’s. In medical imaging, the field’s purpose is to align the spin of protons—the nuclei of hydrogen atoms—in the body. (A spinning proton acts as a sort of bar magnet; like a compass needle, it points in the direction of a surrounding field.)
Tucked inside the big magnet is the RF coil, which transmits radio-frequency waves. When the frequency of a wave pulse matches the spin rate of the protons, the spin “flips” direction by 90 degrees. When the pulse ends, the protons relax back to their original alignment, a lower energy state. The lost energy departs as a radio signal, which is picked up by a receiver. The density of signals gives information about the molecular makeup of bodily tissues—distinguishing bone from blood, white matter from gray matter, tumors from healthy tissue.
Knowing where in the body the signals originate requires yet another set of coils. Sandwiched between the main magnet and the RF coil, the gradient coils generate a magnetic field that makes the main field stronger in some places and weaker in others. This variation changes the frequency of the protons’ signals depending on their location in the field, which allows a computer to calculate their location in the body. By pulsing the gradient and RF coils on and off in various configurations, the machine produces a three-dimensional picture.
It’s the gradient field that typically makes metal objects problematic in an MRI machine. Because the magnetic force the field exerts is uneven, it can slingshot an object through the body. That’s why people with pacemakers or lodged bits of shrapnel don’t get MRIs. MINIBOTS FOR MEDICAL MISSIONS
Across the globe, researchers are engineering a wide array of free-roaming robots to carry out precise and delicate tasks inside the human body. Here are some of the most innovative designs. Illustrations: James Provost
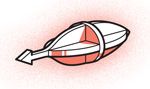
Steerable Surgeons
Lead engineer: Brad Nelson, Swiss Federal Institute of Technology Zurich
Body design: Flat nickel parts are assembled to make a 3-D tool.
Power source: External electromagnetic coils
Steering mechanism: Magnetic field gradients
Tracking device: Microscope
Size (length): 1 millimeter
Applications: Retinal surgeries, drug therapy for ocular disease
Status: Navigation has been tested in rabbit eyes.
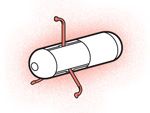
Robot Pills
Lead engineer: Paolo Dario, Scuola Superiore Sant’Anna, Pisa, Italy
Body design: A capsule contains a magnet, camera, wireless chip, and set of mechanical legs.
Power source: DC motors, magnets outside the body
Steering mechanism: Magnetic field gradients
Tracking device: Camera, wireless telemetry system
Size (length): 2 centimeters
Applications: Disease screening
Status: Visual inspection has been tested in a pig’s intestines.

Plaque Busters
Lead engineer: Jong-Oh Park, Chonnam National University, South Korea
Body design: A magnetic capsule is equipped with a micro drill head.
Power source: Electromagnetic coils
Steering mechanism: Magnetic field gradients
Tracking device: MRI and X-ray imaging
Size (length): 10 millimeters
Application: Removal of plaque in the arteries
Status: Prototype is being developed.
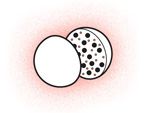
Magnetic Microcarriers
Lead engineer: Sylvain Martel, École Polytechnique de Montréal
Body design: Magnetic nanoparticles and anticancer drugs are encased in a biodegradable polymer sac.
Power source: MRI machine
Steering mechanism: Magnetic field gradients
Tracking device: MRI imaging
Size (diameter): 50 micrometers
Application: Anticancer drug delivery
Status: Navigation and drug release have been tested in rabbits.
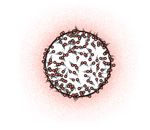
BacteriaBots
Lead engineer: Metin Sitti, Carnegie Mellon University, Pittsburgh
Body design: Swarms of bacteria self-attach to polymer beads.
Power source: Flagella of living bacteria
Steering mechanism: Chemical, gas, temperature, or acidity gradients
Tracking device: Fluorescent or X-ray imaging
Size (diameter): 55 micrometers
Application: Disease diagnosis and drug delivery
Status: Chemical navigation has been tested in the lab.
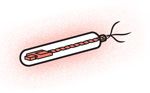
Corkscrew Swimmers
Lead engineer: James Friend, Royal Melbourne Institute of Technology, Australia
Body design: A capsule contains a piezoelectric rotary motor that whips an attached tail.
Power source: Wireless power transmission
Steering mechanism: Piezoelectric vibrations
Tracking device: X-ray imaging
Size (diameter): 250 micrometers
Application: Vessel navigation
Status: Motor has been tested using applied electric currents in the lab.
But there’s a way to get around the projectile problem. Consider the pig experiment. First, as the metal bead slips through the catheter into the artery, only the main magnet is turned on. Its field strongly magnetizes the bead, but because the field is uniform, the bead doesn’t move. In this “supermagnetized” state, a slight tug from the gradient coils will nudge the bead forward without increasing its magnetization. This prevents the bead from catapulting toward the source of the pull—as it would in the presence of a permanent magnet—and allows us to control its acceleration. It also lets us reduce the size of the bead and still move it anywhere in the body using relatively weak gradients. Then, by shifting the direction of the gradient field with precise algorithmic instructions, we can steer the bead through the artery.
The pig experiment proved the concept, but a naked 1.5-millimeter metal bead isn’t much use for transporting drugs inside the microvasculature of the human body. To make a real medical microbot, we started with iron cobalt nanoparticles, which are more sensitive to magnetic forces than other nontoxic metal alloys. We coated the nanoparticles in graphite to keep them from oxidizing, and then we encased them, along with molecules of the cancer drug doxorubicin, in a biodegradable polymer sac.
We can scale these robots down to a few hundred micrometers; our gradient coils aren’t powerful enough to get a grip on anything smaller than that. With beefier, custom-built coils, we reduced the size to about 50 µm. But that’s the limit: Propelling even tinier robots would require gradients so strong that the huge jolt of current needed to quickly fire up the coils would begin to disturb cells in the body’s central nervous system.
In 2011, we tested the ability of these microcarriers to deliver drugs in rabbits. We injected a small squad into the hepatic artery, which supplies blood to the liver and other nearby organs. As in the pig experiment, we had already plotted a simple navigation course using an MRI scan of the rabbit’s interior. Our computer program then steered the robots toward their target by automatically adjusting the gradient field. We waited a few seconds, then took another scan. A distinguishable black smudge in one lobe of the liver proved that the tiny soldiers had shot through the artery, made a sharp turn at a bifurcation, and then congregated at the last waypoint. As their polymer shells degraded, they released their medicinal weapons, although in this experiment, there was no tumor to kill.
Microcarriers may be ideal for targeting liver cancers in humans because the vessel branches that lead to the liver’s lobes are quite large—about 150 µm or wider. But to reach tumors hidden behind networks of smaller capillaries, such as in the breast or the colon, we need microbots no thicker than a couple of micrometers. And because the gradient coils of an MRI machine can’t be made powerful enough to propel them, they must propel themselves.
We don’t have to invent these dream machines—they already exist in nature.
The MC-1 bacterium was first discovered in 1993 in a small, salty estuary of the Pettaquamscutt River in Rhode Island. The strain, which is now cultivated in laboratories, is the model microbot. It has a spherical body about 2 µm across, which provides ample surface area for attaching molecules of anticancer drugs. It also has a pair of spinning, whiplike tails powered by molecular “rotary motors” that rocket it through water at speeds up to 150 times its body length per second. (If Olympian Michael Phelps could do that, he would swim faster than the cruising speed of a Boeing 747.)
Most important, MC-1 is magnetic. The species belongs to a group of bacteria described as magnetotactic because they have the special skill of aligning themselves with Earth’s magnetic field. Though it’s unclear what advantage this gives them, they perform the trick with a chain of iron oxide nanocrystals called magnetosomes that behave like a compass needle.
To guide the bacteria, we need generate only a very weak magnetic field—just slightly stronger than Earth’s—pointed toward a target such as a tumor. The goal here is to direct the compass needle (the magnetosomes), not to propel the entire compass (the organism). We do this with specially designed electromagnetic coils that, in addition to being less powerful than an MRI machine, allow us to orient the field by varying the amount of current passing through each coil. The bacteria then swim toward the artificial “north” pole, navigating the twists and turns of the vascular network as if they were reeds in a river. In the hot blood of a mammalian body, the microbes survive only about 40 minutes.
We have demonstrated the potential of this system in mice. After injecting a swarm of bacteria in each animal’s tail, we fixed an artificial pole on one of two tumors in the body. When we dissected and examined the tumors, we found more bacteria in the targeted tumor than in the other. But the results weren’t ideal. In fact, most of the swarm never made it to a tumor at all. Many cells got swept away by heavy currents in the large vessels of the tail before they could reach the calmer capillaries.
I offer two solutions to this problem. First, swarms of MC-1 bacteria could be used specifically to treat colorectal cancers. Because the colon is easily accessed through the rectum with a catheter, the bacteria can be injected close to the tumor and so will be more likely to find it. We are now planning trials in monkeys to test whether the procedure will be safe for humans.
The second solution we are pursuing is a microcarrier-bacteria hybrid [see illustration, “Fantastic Voyage”]. We make these robots by loading fat-like bubbles, such as vesicles or micelles, with magnetic nanoparticles and drug-packing bacteria. When deployed in the body, they could be navigated using an MRI machine through large blood vessels, similarly to microcarriers, until narrower vessels block their path. At this point, the bubbles—which could be engineered to burst upon smacking into a vessel wall—would dispatch the bacteria. Then the patient would be transferred from the MRI machine into our custom coils. Guided by the coils’ magnetic field, the armed microbes would swim to the tumor. And there they would die, releasing their drug cargo.
It will be several years before medical microbots of any form will be used in patients. There are still many unanswered questions. How many robots are needed to deliver enough drugs to kill a tumor? What amount of bacteria is safe to inject in humans? Will the body raise antibodies against them?
Luckily, we aren’t alone in tackling these challenges. The field is growing; roboticists around the world are inventing a whole family of tiny devices for various tasks [see sidebar, “Minibots for Medical Missions”]. There is no shortage of possibilities. In my laboratory, we have already started to investigate ways of exploiting the energy properties of magnetic microbots to temporarily open up the blood-brain barrier and access tumors in the brain. Almost 50 years after Hollywood dreamed up the tale, a real-life fantastic voyage is just beginning.
This article originally appeared in print as “Journey to the Center of A Tumor.”
About the Author

Sylvain Martel founded and directs the NanoRobotics Laboratory at École Polytechnique de Montréal. He is also a pioneer in the burgeoning field of medical microrobotic.
Sylvain has published a book on “A Microscopic Submarine in My Blood” on 24 August 2016 and it will be exactly 50 years after the release of the movie Fantastic Voyage. This will also correspond to the date where the first interventional nanorobotic facility for cancer therapy enabling the same scenario as in the Fantastic Voyage movie to reach the brain through the vascular network would be done with medical staff and engineers.
A Microscopic Submarine in My Blood



 Ascertained and Presumed ‘Truths’: A Discussion about Claiming and Denying
Ascertained and Presumed ‘Truths’: A Discussion about Claiming and Denying